Review Article - (2023) Volume 14, Issue 6
Abstract
Background: Chronic diseases account for about 59% of the 56.5 million deaths reported worldwide and 46% of the global burden of the disease. Nearly half of deaths from chronic disease are attributed to cardiovascular disease, obesity, and diabetes. The present review will give an outline about the role of essential nutrients on cardiovascular health, chemistry, dose, drug metabolism and pharmacokinetics, mechanism of action, risk, and discuss the evidence-based risk and management of drug interaction. The information was collected from books, and electronic search (PubMed, Science Direct, Lilca and Scielo) and PRISMA.
Discussion: Nutrient in the form of pharmaceuticals available can be used for the prevention and treatment of cardiac diseases. Adverse drug reactions, nutrients- drug interactions, and iatrogenic diseases have been identified as significant factors responsible for patient morbidity and mortality. A better understanding of these mechanisms and recent developments in laboratory technology can help assess possible drug interactions when drugs are prescribed at the same time. Increased knowledge of inter-individual variation in drug decomposition capacity and recent results related to nutrient and nutraceutical influence can be used to reduce adverse drug reactions and disease iatrogenic.
Conclusion: There is a need to enhance and foster interdisciplinary communication between medical herbalists, physicians, and dieticians. According to dieticians food may interact with conventional drugs and that drugs may affect nutritional status, in order to provide the patient with proper dietary suggestions, and to allow the maximum effectiveness and safety of drug therapy, while preserving/correcting the nutritional status.
Keywords
Food, Supplements, Cardiovascular drugs, Interactions, Risk management
Introduction
Diet and nutrition are important factors in the promotion and maintenance of good health throughout life and also play as a risk factor for chronic diseases (Matsudo V, et al., 2002). As per calculations, in 2001, chronic diseases contributed approximately 60% of the 56.5 million total reported deaths in the world contributed by chronic diseases and approximately 46% of the global burden of disease (WHO, 2002). The share of the NCD burden is expected to reach 57% by 2020. Nearly half of deaths from chronic diseases are caused by cardiovascular disease. The 2016 Heart Disease and Stroke Statistics update of the AHA reported that the overall death rate from CHD was 102.6 per 100,000 (Mozaffarian D, et al., 2016). There is a strong relationship between SCD and CHD (Montagnana M, et al., 2008). Clinical and post-mortem studies as well as data from death certificates revealed that 62%-85% of patients who suffer out-of-hospital SCD have evidence of prior CHD, 10% have other structural cardiac abnormalities, and 5% have no structural cardiac abnormality (Kannel WB and Thomas Jr HE, 1982; Zheng ZJ, et al., 2001). A surveillance study of SCD from Ireland concluded that the majority of cases occurred at home and that successful resuscitation of SCD was especially associated with ventricular fibrillation as presenting rhythm (Byrne R, et al., 2008). IHD was also the greatest single cause of death in 2000, accounting for an estimated 6.0 million deaths. Individual populations face different challenges and each population has unique health burdens, however, CVD remains one of the greatest health challenges both nationally and worldwide (McAloon CJ, et al., 2016). Obesity is associated with some of the major risk factors for CVD, such as hypertension and low concentrations of High-Density Lipoprotein-cholesterol (HDL-cholesterol) (WHO, 2002). Arterial blood pressure and Hypercholesterolemia are key factors in the development of CVD.
Literature Review
Extensive study has been done in order to derive the solution to the question at hand. The majority of the study conducted revolves around the usage of e-books, most of which focused exclusively on essential nutrients. Additionally, government websites provided more information regarding what types of medications are used on cardiovascular diseases, as well as the short-term and long-term effects of their use. Furthermore, a couple of case studies detailed some students in India, and how their health improved with a conversion to a essential nutrients. The sources that are being used are several academic, peer-reviewed research papers, books, journals, and case studies. The mode of methodology that will be used to carry out the research is mixed between data analysis and experiments given in the case studies.
There has been a boom in their sales as patients rush to self-medicate, either in the hope that these products will be effective in treating diseases unsatisfactorily treated with pharmaceuticals, or that the adverse effects of some pharmaceuticals may be avoided. On whole, ‘Nutraceuticals’ has led to the new era of medicine and health, in which the food industry has become a research-oriented sector and that is used for the improvement of health, by preventing or treating disease (Das L, et al., 2012). The relation between nutraceuticals and other health products for various diseases are presented in Figure 1.
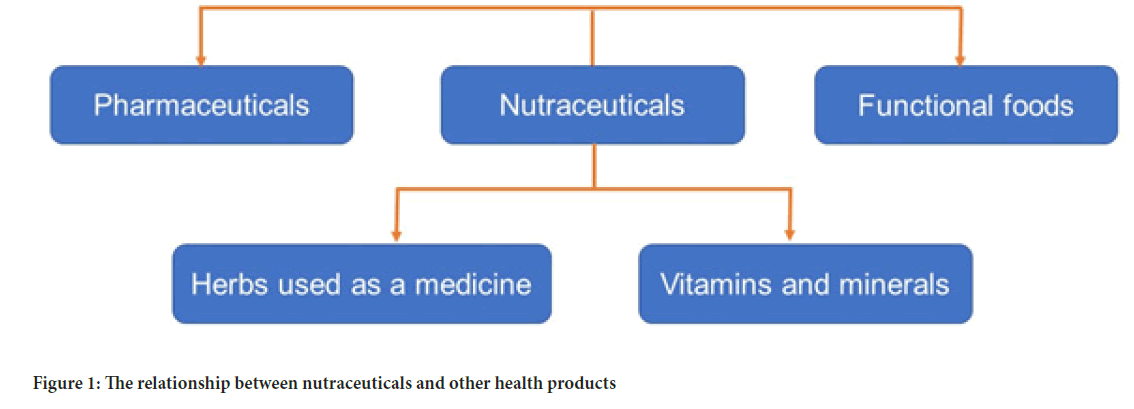
Figure 1: The relationship between nutraceuticals and other health products
Nutraceuticals in the form of macronutrients, micronutrients algae, and herbs are recommended together with physical exercise for prevention and treatment of CVD (Muredzi ED, 2023). Table 1presents some of the more recognizable nutraceuticals substances grouped according to food-source providers (Wildman RE, 2016).
| Plants | Animals | Microbials | |
|---|---|---|---|
| Ascorbic acid | Potassium | Eicosapentaenoic Acid (EPA) | Bifidobacterium bifidum |
| β-glucan | Glutathione | Conjugated Linoleic Acid (CLA) | Saccharomyces boulardii (yeast) |
| Cellulose | Lycopene | Lecithin | L. acidophilus (NCFB 1748) |
| Daidzein | β-carotene | Minerals | |
| Gallic acid | Lignin | Coenzyme Q10 | |
| Indole-3-carbinol | Geraniol | Zinc | |
| Luteolin | Genistein | Choline | Lactobacillus acidophilus (LC1) |
| Lutein | Hemicellulose | Calcium | Streptococcus salvarius (S. thermophilus) |
| Perillyl alcohol | Capsaicin | Selenium | |
| Pectin | α-Tocopherol | Creatine | |
| Quercetin | δ-Limonene | Spingolipids | B. infantis |
| Selenium | Nordihydrocapsaicin | B. longum | |
| γ-Tocotrienol | Allicin | DocosaHexenoic Acid (DHA) | |
Table 1: Examples of nutraceutical substances grouped by food source
Discussion
Risk of cardiovascular disease
Cardiovascular disease is the leading cause of premature morbidity and mortality in industrialized countries. Serum cholesterol correlates with the risk of coronary artery disease, myocardial infarction, and coronary death (Anderson KM, et al., 1987). Several studies have addressed the relation between the intake of Polyunsaturated Fatty Acids (PUFAs) and cardiovascular disease and the effect of increased PUFA intake on cardiovascular events. The low mortality from cardiovascular disease among traditionally living Eskimos and Alaskan Natives has been attributed to less atherosclerosis in the coronary arteries (Middaugh JP, 1990). The factors affecting the risk of heart disease are summarized in Figure 2(Dominguez H, 2013).
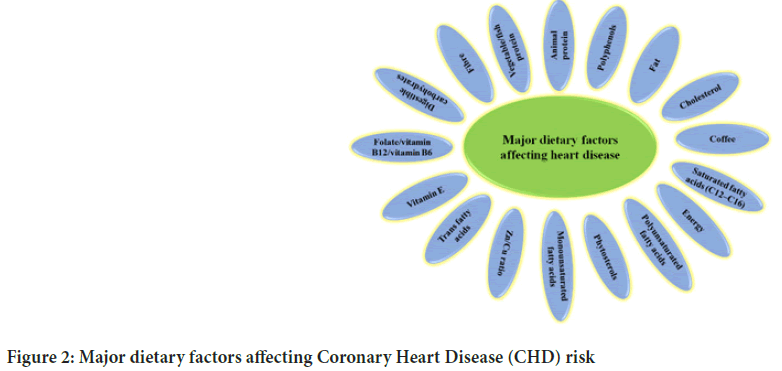
Figure 2: Major dietary factors affecting Coronary Heart Disease (CHD) risk
Nutraceutical diet for better cardiovascular health
Several nutraceutical substances are found in higher concentrations in specific foods or food families. These include capsaicinoids, which are found primarily in pepper fruit, and allyl sulfur (organo sulfur) compounds, which are particularly concentrated in onions and garlic. The table provides a listing of certain nutraceuticals that are considered unique to certain foods or food families (Wildman RE, 2016). Major Dietary factors are known to influence cardiovascular risk (Table 2) (Dominguez H, 2013).
| Factors | TC | LDLc | HDLc | TG | BP | Aggregation | ED | Oxidation | Others |
|---|---|---|---|---|---|---|---|---|---|
| Energy | ↑↑ | - | ↑ | ↓ | - | - | ↓ | - | ↑ Bodyweight |
| Fat | ↑↑ | ↑ | ↑ | - | - | - | ↓ | - | ↑ Factor VII |
| Saturated fatty acids (C12-C16) | ↑↑↑ | ↑↑↑ | ↑ | - | ↑ | ↑ | ↑ | ↓↓ | ↑ Insulin resistance |
| Monounsaturated fatty acids | ↓↓ | ↓↓ | ↑ | - | ↓ | - | ↓ | ↓? | ↓ Fibrinolysis; ↓ PAI |
| n-6 polyunsaturated fatty acids | ↓↓↓ | ↓↓↓ | ↓↓ | - | - | - | - | - | ↑ |
| n-3 polyunsaturated fatty acids | ↓? | ↑ | ↓↓ | ↓↓ | ↓ | ↓ | ↓? | - | |
| Trans fatty acids | ↑ | ↑ | ↓ | ↑ | ↑ | ↑? | - | - | ↑ Lp(a); ↑ Insulin resistance |
| Cholesterol | ↑ | ↑ | ↑W | - | - | - | - | - | - |
| Alcohol* | ↓ | ↓ | ↑ | ↓ | ↓ | ↓ | ↓ | ↓↓ | Various effects |
| Digestible carbohydrates | ↓ | ↑↑** | - | - | ↓ | ↓↓ | **Insulin resistance | ||
| Vegetable/fish protein | ↓ | ↑ | ↓ | - | - | ↓ | - | ↑ (NO) | |
| Animal protein | ↑ | ↑ | - | - | - | ↓ | - | - | |
| Fiber | ↓ | ↓ | - | - | ↓ | - | ↓ | - | - |
| Phytosterols | ↓↓ | ↓↓ | - | - | - | - | ↓ | ↓? | ↓ Antioxidants |
| Folate/vitamin B12/vitamin B6 |
- | - | - | - | - | ↓? | ↓ | - | ↑ Homocysteine |
| Vitamin E | ↓? | - | - | - | - | ↓ | ↓ | ↓ | - |
| Ca | ↓ | - | - | - | ↓ | - | - | - | - |
| Zn/Cu ratio | ↑ | ↑ | - | - | - | - | - | - | - |
| Polyphenols* | ↓ | ↓ | ↑ | ↓ | ↓ | ↓ | ↓ | ↓↓ | Most information in vitro |
| Coffee | ↑↑ *** | ↑↑*** | - | ↑↑ *** | - | - | - | ↓↓ | - |
Note: TC: Total Cholesterol; LDLc: Low-Density Lipoprotein Cholesterol; HDLc: High-Density Lipoprotein Cholesterol; TG: Triglycerides; BP: Blood
Pressure
↑: Increase; ↑↑: Abnormal increase; ↓: Reduce; ↓↓: Abnormal reduction; ↓? and ↑?: Limited scientific evidence; *: High consumption; w: Effects in
women; **Fructose; ***Cafestol and Kawheol (coffee diterpene)
Table 2: Major dietary factors affecting Coronary Heart Disease (CHD) risk
Essential nutrition required to maintain the heart health is depicted in Figure 3. Minerals exert various effects on CVD. Diets with high zinc/copper ratios are hypercholesterolemia (Bastida S, et al., 2000). Some minerals play a central role in blood pressure regulation; however, apart from sodium or potassium, little is known about the effects of minerals on CVD. Increased intakes of magnesium and calcium decrease blood pressure and the risk of being hypertensive (Rodenas S, et al., 2011).

Figure 3: Essential nutrients for cardiovascular disease
Sodium: A modest reduction (9-12 to 5-6 g/day) in salt intake for four or more weeks causes significant falls in blood pressure in both hypertensive and normotensive individuals. Reduction in salt intake of 3 g/day would result in the reduction of incidence CHD (5.9%-9.6%), stroke (5.0%-7.8%) (Bibbins-Domingo K, et al., 2010).
Potassium: The risk of stroke varies greatly with potassium intake: The relative risk of stroke in the highest quintile of potassium intake (average of 110 mmol/day) is almost 40% lower (He FJ and MacGregor GA, 2001).
Vitamin A: It is a liposoluble compound derived from animal tissues. Retinal, retinoic acid and β-carotene have potential activity against atherosclerosis and inflammatory diseases also have antioxidant activity (Walden R and Tomlinson B, 2012). Consumption of carotenoid-rich vegetables has been linked to a reduced risk of coronary artery disease. An oxygenated carotenoid (Lycopene) whose antioxidant activity is two times higher than that of β-carotene. Tomatoes are the best source of lycopene (Sesso HD, et al., 2012).
Vitamin C: The population with vitamin C deficiency, found at high risk for coronary heart disease (Simon JA, 1992). Antioxidant properties of Vitamin C may synergistically act with vitamin E, this leads to a reduction in peroxyl radicals’ formation and lipid peroxidation blocking (Mikirova NA, et al., 2008). Citrus extract plus ascorbate strongly inhibited atherosclerosis (Vinson JA, et al., 1998). Plant flavonoids are powerful antioxidants for heart disease (Vinson JA, et al., 1995). Studies have shown a reverse relationship between plasma renin activity levels and vitamin D, coronary artery calcification, and hypertension, (Resnick LM, et al., 1986).
Vitamin E: Vitamin E antioxidant and anticoagulant properties stabilized plaque, platelet aggregation, reduced inflammation, enhancing vasodilation, expression of adhesion molecules on the arterial wall are the potential mechanisms of cardio-protection (MI and thrombotic stroke) with vitamin E (Meagher EA, et al., 2001). High fiber consumption reduces post prandial glucose responses after carbohydrate-rich meals and lowers total and LDL cholesterol levels (Weickert MO and Pfeiffer AF, 2008). The polyphenolic contentis used for CVD prevention (Ullah MF and Khan MW, 2008). Red wine (polyphenolic) taken before food increases serum antioxidant activity which activates the platelet NO synthase and inhibits the platelet activation and production of reactive oxygen species, for at least 4 hours (Maxwell S, et al., 1994). Sudden death and heart attack incidences decreased by consumption of flavonoids (luteolin, kaempferol, apigenin, myricetin, and quercetin) intake assessed by dietary history. Flavonoid rich apples, black tea and onions were considered the most suitable foods. Moderate consumption of wine was found beneficial on ischemic CVD (Gresele P, et al., 2008). Resveratrol, induces the expression of several longevity genes, and prevents aging-related decline in cardiovascular function (Das DK, et al., 2011).
Flavanol-rich cocoa: They are suggested to have anti-inflammatory properties, antioxidant properties, antihypertensive effects, and anti-platelet aggregation. Platelet reactivity reduces and coronary vasomotion improved by Dark chocolate. It was found a significant decrease in the risk of the combined outcome of first MI or stroke in people in the top quartile compared to those in the bottom quartile of chocolate consumption (Buijsse B, et al., 2010). It seemed to be partly due to a drop in blood pressure in people who consume more chocolate. Vitamin K deficiency in the diet has been linked to an increased risk of atherosclerosis and soft-tissue calcification (Erkkilä AT and Booth SL, 2008). High level of potassium, magnesium, and calcium can lead to lower blood pressure and lower CDA and stroke (Houston MC and Harper KJ, 2008). The multiple cardio-protective and physiological activities of magnesium include calcium channel-blocking effects, antiarrhythmic effects, inhibition of blood coagulation, and improvement in NO release from coronary endothelium (Shechter M, et al., 1999). However, potassium and magnesium need to be avoided in the event of kidney failure. The health significance of iron is well known, with increasing attention being paid to the identification and treatment of iron deficiency in heart failure. Anemic patients (Hb ≤ 12 g/dL) with stable CHF, intravenous injections of iron sucrose only for 12 days decreased symptoms, improved exercise capacity, and increased hemoglobin (Anker SD, et al., 2009).Trace minerals
Chromium: Chromium is an essential mineral that appears to have a beneficial role in the regulation of insulin action, metabolic syndrome, and cardiovascular disease (Hummel M, et al., 2007). Chromium is important for the metabolism of glucose and lipids and can contribute to the regression of cholesterol-induced atherosclerosis. The deficiency of chromium may influence glucose and lipid metabolism and affect atherosclerosis (Schroeder HA, 1967). Insulin resistance can be an important factor in disrupting lipid metabolism, the favorable effect of chromium on glucose or insulin metabolism can be a key factor in improving lipid profile (Balk EM, et al., 2007). Insufficient selenium in the soil can cause cardiac arrhythmias, CHF, multifocal myocardial necrosis, with cardiomyopathy. Primary prevention trials and most secondary prevention studies suggest that supplementation does not significantly reduce ischemic CVD events (Hercberg S, et al., 2004). L-carnitine was used as a supplemental treatment for Percutaneous Coronary Intervention (PCI) for Non-ST Elevation Acute Coronary Syndrome (NSTEMI).
DHA and EPA can improve many CVD risk factors, including lowering of plasma triglycerides, blood pressure, platelet aggregation, and inflammation, and improvement of vascular reactivity at the dose of >3 g/day (Breslow JL, 2006). A link has been established between fish oil supplementation and a significant reduction in deaths due to heart causes (León H, et al., 2008). Coenzyme Q10 (ubiquinone) has been used in oral forms to treat various cardiovascular disorders including angina pectoris, hypertension, and CHF (Greenberg ER, et al., 1996).
Algae and cardiovascular health
The importance of algae as components of functional foods with special application to cardiovascular diseases, due to their high dietary fiber, mineral, vitamins, and phytochemical contents, and their polyunsaturated fatty acid. Effects of seaweeds and alga compounds on cholesterol metabolism, lipoprotein levels, and thrombosis/blood coagulation have presented in Table 3.
| Seaweeds and alga compounds | Source and content | Bioactivity | References |
|---|---|---|---|
| Whole algae orlipids extracts (unicellular alga Nannochloropsis sp.) | DHA-enriched oils from algae | Reduce the plasma and liver cholesterol levels in male rats | Werman MJ, et al., 2003 |
| Polysaccharides and alginates | Sargassum horneri, Different red and brown seaweeds |
|
Athukorala Y, et al., 2009 |
| Amino sugar-containing fucansulfate | Ecklonia kurome, Fucus sp. | Anticoagulant and antithrombin activities | Nishino T, et al., 1994 |
| Brown seaweed fucans | Ascophyllum nodosum | Anticoagulant | Chevolot L, et al., 1999 |
| Marine algae | Undaria pinnatifida (Wakame) |
|
Murata M and Nakazoe JI, 2001 |
| Heparinoid-active sulphated polysaccharides | Laminaria hyperborea | Anticoagulant (sulfate-ester form) | Shanmugam M and Mody KH, 2009 |
| Porphyran | Nori (The dried alga contains protein, ash, vitamins, and carbohydrate+Zn, Cu, Mn, and Se) |
|
Noda H, 1993 |
| Dipeptides | Undaria pinnatifida | Antihypertensive | Sato M, et al., 2002 |
Table 3: Effect of seaweeds and alga compounds on cholesterol metabolism, lipoprotein levels, and thrombosis/blood coagulation
There has been a combined effort among scientists to explore and utilize varying food sources to develop functional foods to cater to the ever-increasing demand from the consumers, who seek health-promoting roles of dietary compounds (Mendis E and Kim SK, 2011). Marine organisms are considered safer alternatives to some existing synthetic drugs as they exhibit significant biological properties. Marine bioactive include peptides, fatty acids, carbohydrates especially Sulfated Polysaccharides (SPs). Time is useful in food industries and performs the healing junctions like anti-hyperlipidemic, antioxidant, anti-diabetic, and these all functions contribute towards cardiac protection (Tufail T, et al., 2018). Nutrient used to prevent the risk of heart diseases are summarized in Table 4. Interaction between essential nutrient and conventional cardiovascular drugs are briefly summarized in Table 5and graphically presented in Figure 4.
| Nutrients and their source | Chemistry and dose | Metabolism and pharmacokinetics | Therapeutic areas and use | References | |||
|---|---|---|---|---|---|---|---|
| 1. Tea Source-Camellia sinensis (top two leaves and the bud) |
|
Tea polyphenols have low bioavailability due to their high molecular weights and a high number of hydroxyl substituents. The hydroxyl groups may hinder the absorption of the compounds across the gut lumen. | Cardiovascular, bone, skin and oral health, cancer prevention and weight management (antioxidant) | Mukhtar H and Ahmad N, 2000 | |||
| 2. Soy (Isoflavones) Source-Whole soybeans, soy sauce, tofu (soybean curd) |
|
In healthy individuals, an extract of soy did not induce the cytochrome P450 isoenzyme CYP3A49. In vitro, soybean products and an extract of hydrolyzed soybean, as well as soybean isoflavones genistein and daidzein, inhibited CYP2C9, and CYP3A49 | Cardiovascular, mental, bone, women’s and skin health, cancer prevention (antioxidant and estrogenic) | Dewell A, et al., 2006 | |||
| 3. Flaxseed Source-Linum usitatissimum L. (Linaceae) |
|
Administration of 10 g flaxseed to humans resulted in large increases in fecal lignan excretion, from 727 to 12,871 nmol/day. | Cardiovascular health, cancer prevention, women’s health. (antioxidant and weakly oestrogenic) |
Rowland I, et al., 2003 | |||
| 4. Coenzyme Q10 Source-(Fatty fish, cereals, poultry, and vegetables, |
|
In the gastrointestinal tract, the uptake of coenzyme Q10 is relatively slow and is dependent on postprandial. It is present in human plasma at a level of 1 mg/L.11 | Cardiovascular health, cancer prevention, respiratory, skin and animal health (antioxidant) | Weber C, et al., 1997 | |||
| 5. Creatine Source-Synthesized in the kidney, liver, and pancreas |
|
Plasma level of creatine is maximum within 2 hrs after ingestion of dose under 10 g but it takes 3 hrs or more when ingested more than 10 g. Clearance depends on both skeletal muscle and renal function. | Heart and neuromotor disorder, muscular dystrophy, Parkinson’s disease, and Huntington’s disease | Persky AM and Brazeau GA, 2001 | |||
| 6. Carnitine Source-Synthesized from lysine and methionine in the liver and kidney |
|
Bioavailability: 54%-87%, depending on a food level, and the bioavailability from supplements (0.5-0.6 g) has been estimated to be 14%-18% of the dose. | Sports enhancement, cardiovascular and bone health, weight optimization, veterinary health | Kletzmayr J, et al., 1999 | |||
| 7. Lycopene Source-A natural red pigment found in plants, fruits, and vegetables like tomatoes |
|
Plasma concentrations ranging from 0.22 to1.06 µmol/L. After administration of tomato juice which contains high level of lycopene, peak plasma concentrations were reported from 24 to 48 hours, and elimination half-life was estimated to be 48-72 hours. |
Cardiovascular disease and cancer prevention, especially prostate cancer. | Diwadkar-Navsariwala V, et al., 2003 | |||
| 8. Lutein source-Present in green vegetables, particularly spinach) |
|
The bioavailability depends upon the chemical and physical nature of the food source, and co-consumed material, particularly the fat content. | Cardiovascular, eye, and skin health (antioxidant) | Alves-Rodrigues A and Shao A, 2004 | |||
| 9. Dehydroepiandrosterone Source-outer layer of the adrenal glands |
|
It is an endogenous hormone synthesized and excreted mainly in the adrenal gland. The elimination half-life of DHEA is 15-38 minutes, and for DHEAS it is 7-22 hours. Oral absorption is excellent. | Cardiovascular and mental health, veterinary health | Pepping J, 2000 | |||
| 10. Policosanol/Octacosanol Source-Superficial fruit, leaves, and whole grains |
|
Absorption of octacosanol has been ‘assumed’ to range between 10% and 35%, and bioavailability ranges between 5% and 12%. It is unlikely that policosanol will metabolize or inhibit other drugs that are substrates of liver enzymes. | Cardiovascular disorders and also used in Parkinson’s disease, and for enhancing athletic performance. | Lin Y, et al., 2004 | |||
| 11. Pycnogenol Source-Bark of Pinus pinaster |
|
Metabolites of procyanidins were identified after oral intake by humans, and maximum excretion was seen after between 8 and 15 hours, indicating slow absorption and metabolism. | Cardiovascular, eye, respiratory, and oral health (antioxidant) | Düweler KG and Rohdewald P, 2000 | |||
| 12. Melatonin Source-Bananas, tomatoes, cucumbers, and beetroots |
|
Oral doses of melatonin have a short half-life and are quickly cleared, and even very high nightly doses of 50 mg are cleared by the following morning However, after two weeks of high daily dosing, lipid storage occurs. | Cardiovascular health, cancer prevention, sports enhancement, sleep improvement, and bone health (antioxidant) | - | |||
| The legal classification of melatonin: In the UK, the Medicines and Healthcare Products Regulatory Agency (MHRA) has restricted to prescribe melatonin, available on a named patient basis only. There are no licensed products in the UK, so it is unlawful to promote melatonin. However, in the USA it may be sold as a food supplement, under the Dietary Supplement Health and Education Act of 1994. British residents can legally bring melatonin purchased in the USA home, for personal use. | |||||||
| 13. Resveratrol Source-Leaves, skins and petals of Vitis vinifera, and also in wines and grape juice |
|
After oral administration of 25 mg resveratrol to humans, the highest plasma concentrations were detected after 30 minutes, which returned to baseline after 2 hours. In other studies, the effect of resveratrol on CYP1A2 was very small. | Anti-aging effects, antioxidant, oestrogenic, antiplatelet effects, cardiovascular diseases, including atherosclerosis. | Wolter F, and Stein J, 2002 | |||
| 14. Grape seed pro-anthocyanidin Source-Extract of Vitis vinifera L. (Vitaceae) |
|
The pro-anthocyanidins are poorly absorbed in the small intestine, and it is thought that ingestion results in metabolism by colonic bacteria. 3-Hydroxy-phenylpropionic acid has been identified as the major metabolite. | Promotes general cardiovascular health and skin health (antioxidant) |
Ward NC, et al., 2004 | |||
Table 4: Nutritional summary for prevention of heart disease risk
| Nutrients+drug | Dose | Evidence-based drug interaction | References |
|---|---|---|---|
| Tea+alprazolam | Alprazolam 2 mg dose before and after decaffeinated supergreen tea extract. |
|
Donovan JL, et al., 2004 |
| Tea+antiplatelets | Tea along with aspirin of 325 mg daily | No significant effect on platelet aggregation | Hodgson JM, et al., 2002 |
| Tea+buspirone | 4 capsules of green tea catechin (tea flavonoids) extract daily for 28 days | 21% AUC reduced with a single dose of 10 mg buspirone | Chow HS, et al., 2006 |
| Tea+cyclosporin or tacrolimus | - |
|
Zhong Z, et al., 2006 |
| Tea+flurbiprofen | - |
|
Greenblatt DJ, et al., 2006; Dai YH, et al., 2003 |
| Tea+piperine | Tea+piperine (500 micromoles/L) |
|
Lambert JD, et al., 2004 |
| Tea+irinotecan | - |
|
Mirkov S, et al., 2007 |
| Black tea | Black tea-150 ml, 10 women with (anaemia) and (without anaemia) | ↓ in the absorption of radiolabelled iron by 59% in anaemic women) and by 49% in without anaemic women | |
| Green tea | 37 mg catechins as green tea extract | Reduce iron absorption by 26% | |
| Tea+losartan | Subjects: 42 , 4 capsules for 4 weeks daily of green tea extract+25-mg losartanas | There is no expectation of interaction between decaf green tea extract and losartan. | Zijp IM, et al., 2000 |
| Tea+warfarin | Tea (2 to 4 liters daily for one week) | ↓ INR of warfarin from a range of 3.2 to 3.79 and 1.37 | Parker DL, et al., 2009 |
| Isoflavones+cardiovascular | - | In patients with angina pectoris, minimize activation of platelet surface. This indicates that kudzu may increase the risk of haemorrhage when used with antiplatelets or blood thinners. Caution is warranted for concurrent use. | Shaikh AS, et al., 2020 |
| Isoflavones+digoxin | Biochanin A (100 mg/kg) of a 20 mg/kg dose of digoxin taken orally | ↑ the AUC and maximum serum levels (75%). ↔ a terminal half-life and ↔ in mean residence time of digoxin | Peng SX, et al., 2006 |
| Isoflavones+fexofenadine | Biochanin A (100 mg/kg) | Around 30 % and 57 % of oral bioavailability increased and peak plasma concentration of fexofenadine 20 mg/kg. | Peng SX, et al., 2006 |
| Isoflavones+nicotine | Subjects:7 healthy Japanese (non-smokers) Isoflavone tablet six times daily |
There is around 24% ↓ in nicotine metabolism MOA: the metabolism of nicotine is slightly ↓ by Soya isoflavones due toCYP2A6inhibition. | Nakajima M, et al., 2006 |
| With paclitaxel | Genistein 10 mg/kg (orally) | Paclitaxel AUC is modest ↑. | Li X and Choi JS, 2007 |
| Isoflavones+tamoxifen | Subjects: 17 women with confirmed breast cancer. Daily 200 mg isoflavone |
No ↑ in the tumor over 2 to 6 weeks before the surgery | Messina MJ and Loprinzi CL, 2001 |
| Isoflavones+theophylline | Subjects: 20 healthy non-smoking subjects, pre-treatment. 200 mg daidzein twice daily for 10 days+100 mg single dose |
↑ AUC and half-Life of the disposal from approximately 9 hours to approximately 12 hours. The cytochrome P450 isoenzyme CYP1A2 of theophyllines appears mild to isoflavones. | Monostory K and Vereczkey L, 1996 |
| Flaxseed+anticoagulant | Flaxseed oil-rich diet α-linolenic acid 20.5 g daily for 56 days |
↔ significant effect on prothrombin times, bleeding times, or partial prothrombin times | Prasad K, 2009 |
| Flaxseed+antidiabetics | 68 patients+supplement containing 360 mg of flaxseed lignin | ↔ Effect on blood-lipid profile, 0.1% ↓ of (HbA1c). | Lemay A, et al., 2002 |
| Coenzyme Q10+piperine | Subject: 12 healthy subjects, piperine 5mg (Bioperine) with coenzyme Q10 90 mg. | ↔ in the pharmacokinetics of coenzyme Q10 (AUC and maximum level or time to the maximum level) | (Itagaki S, et al., 2009; Badmaev V, et al., 2000) |
| Coenzyme Q10+warfarin | Coenzyme Q10 100 mg per day 4 weeks in 21 Warfarin stabilized patients | ↔ In the INR or the required dose of warfarin. | Landbo C and Almdal TP, 1998 |
| With aldosterone | Single-dose of coenzyme Q10 | ↑ the sodium reabsorption stimulated | Igarashi T, et al., 1975 |
| Creatine+caffeine | 500 mg/kg creatine supplement and 5 mg/kg caffeine capsules for 3 days daily | Lack benefits of creatine in exercises for the extension of the knee on 4th day. One subject experienced some GIT discomfort during use. | Zhou Q, Chowbay B, 2002; Vandenberghe K, et al., 1996; Steenge GR, et al., 2000 |
| Lycopene+food | 13 healthy+lycopene for 14 days as 300 g tomato soup and 60 g tomato paste. | ↔ in the serum levels of lycopene between a high-fat monounsaturated-fat-enriched diet, and a high-carbohydrate low-fat diet. | Ahuja KD, et al., 2003 |
| Lycopene+beta-carotene | A single dose of beta-carotene of 60 mg with lycopene (10 healthy subjects) | In comparison to lycopene administered alone, to significantly improve lycopene AUC by about 4-fold. | Johnson EJ, et al., 1997 |
| With probucol | Lycopene + 500 mg probucol two times | Serum lycopene levels were further reduced by 30%. | Elinder LS, et al., 1995 |
| Lycopene+sucrose polyesters | 194 healthy subjects, the+Olestra 18 g daily | ↓ serum levels of dietary lycopene up to about 30%. It decreases the synthesis of fat-soluble vitamins. | Koonsvitsky BP, et al., 1997 |
| Policosanol+anticoagulants | Policosanol 200 mg/kg+warfarin 200 mg/kg for 3 days in rats. |
|
Carbajal D, et al., 1998 |
| Policosanol+beta blockers | 60 to 80 years age group taking beta-blockers+5 mg policosanol tablets daily | Decrease blood pressure after 1 year from about 141/83 mmHg to 131/81 mmHg, after 3 years decrease to 126/79 mmHg. | Castano G, et al., 2004 |
| With Phenazone | Phenazone 10 mg/kg 25 mg/kg per day. | ↔ Pharmacokinetics | Pérez-Souto N, et al., 1991 |
| Policosanol+nitroprusside | Sodium nitroprusside+200-mg/kg policosanol (pre-treated)as single oral dose | ↑ antiplatelet and hypotensive effect of sodium nitroprusside | Arruzazabala ML, et al., 2001 |
| Melatonin+benzodiazepines | 16 healthy (55 years old), prolonged-release melatonin 2 mg with zolpidem (10 mg) | ↑ impairment of cognitive function. Melatonin alone did not affect cognitive function. ↔ pharmacokinetic | (Otmani S, et al., 2008; Golombek DA, et al., 1992) |
| Melatonin+caffeine | Caffeine significantly ↑the levels of single doses of supplementary melatonin and reduces melatonin synthesis by contesting cytochrome P450 CYP1A2 isoenzyme metabolism. | Obochi GO, et al., 2010 | |
| Melatonin+carbamazepine | Children with epilepsy undergoing carbamazepine monotherapy administered | ↔ Serum levels of carbamazepine and its metabolite. | Gupta M, et al., 2004 |
| Melatonin+nifedipine | Melatonin immediate-release capsules 5 mg per night were given to 47 people with mild. | Systemic and diastolic blood pressure rose moderately (6.5mmHg and 4.9mmHg respectively), and the cardiac rhythm increased (3.9 bpm). | Lusardi P, et al., 2000 |
| Melatonin+oestrogens | In subjects who took a dual oral contraceptive, AUC and a maximum amount of a daily 6 mg dose of melatonin was about 4 times higher | Hilli J, et al., 2008 | |
| Melatonin+propofol | IV propofol+3- or 5-mg+melatonin orally, 100 minutes preoperatively, |
|
Turkistani A, et al., 2007 |
| Melatonin+psoralens | - | Methoxsalen and psoralenic 5-methoxy block melatonin metabolism and increase levels. Increased levels of 5-methoxy psoralen were demonstrated. Psoralens are effective cytochrome P450 CYP1A2 inhibitors. | Souêtre E, et al., 1989 |
| Melatonin+SSRIs | 7 healthy subjects Dose: 40 mg citalopram | No effect on or excretion from the body of endogenous melatonin. | Foster BC, et al., 2015 |
| Melatonin+tobacco | 8 tobacco smokers | AUC of melatonin was almost 3-fold higher. | Ursing C, et al., 2005 |
| Dose: 25-mg dose melatonin | Tobacco smoking ↓ melatonin levels. | ||
| With warfarin | - | Melatonin may ↑ or ↓ the INR in response to warfarin | Herxheimer A, et al., 1996 |
| Resveratrol+aspirin | Resveratrol+50 high-risk cardiac patients taking aspirin | Resveratrol significantly ↓ platelet aggregation | Stef G, et al., 2006 |
| Resveratrol+diclofenac | - | Reveratrol can be addictive with NSAIDs such as diclofenac sodium in some antiplatelet effects. | Yu C, et al., 2003 |
| Resveratrol+mephenytoin | - |
|
Yu C, et al., 2003 |
| With paclitaxel | - | Resveratrol mildly impaired paclitaxel in rats and human liver microsomes. | Václavíková R, et al., 2003 |
| Grapeseed+vit C | Vitamin C alone | ↓ Systolic blood pressure (1.8 mmHg) | Ward NC, et al., 2005 |
| With midazolam | Grapeseed extract+midazolam | No major impacts on midazolam pharmacokinetics. | Nishikawa M, et al., 2004 |
| Grapefruit+fexofenadine | - | For note, the AUC of Fexofenadine is mildly decorated by grapefruit juice and, to a lesser degree, by naringenin, a flavonoid of grapefruit. | Bailey DG, et al., 2007 |
| With calcium-channel blockers | 12 healthy+ homogenised grapefruit extract from the segment free parts+felodipine | The AUC of felodipine ↑ by 3.2-fold (homogenised grapefruit) and 3.6-fold (extract) | Sica DA, 2006 |
| Grapefruit+warfarin | A couple, well stabilized on warfarin, took a few drops of a grapefruit extract. | Hematoma was formed after another 3 days, and its INR was determined to be 7.9 and 5.1 INR (Standard deviation) with no sign of bleeding. | Peynaud D, et al., 2007; Brandin H, et al., 2007 |
Note: ↑: Increase; ↓: Decrease; ↔: No significant change; INR: International Normalised Ratio
Table 5: Descriptive interaction between essential nutrients and cardiovascular drugs
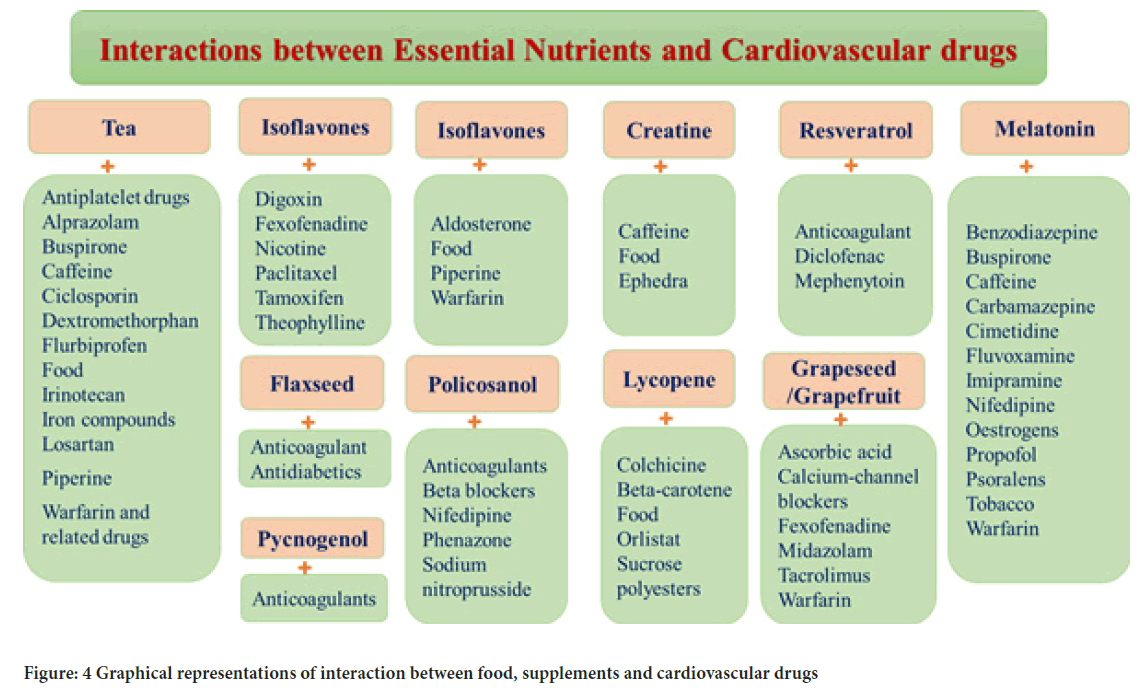
Figure 4: Graphical representations of interaction between food, supplements and cardiovascular drugs
Conclusion
The findings of major clinical trials usually do not support the cardiovascular advantages of antioxidant vitamin or other EMN supplements, and recommendations typically prescribe improved ingestion of foods abundant in these ingredients, not particular additional supplements. Omega-3 PUFAs may be an exception and niacin has an effect on plasma lipids at pharmacological doses. In most cases, scientific test data is not adequate to support conclusive guidelines for nutrients used in CVD prevention or treatment. Concomitant use of nutrients, herbs, and conventional drugs may present with untoward events. The evidence available in the literature indicates various mechanisms through which this can occur. By interacting with conventional medication, nutrients may precipitate manifestations of toxicity or in the other extreme, therapeutic failure. Good knowledge of the potential of commonly consumed nutrients to interact with prescription medicines, irrespective of the nature of the evidence available, will equip health professionals in their practice. Apart from those demonstrated insignificant number of human subjects, not all reported interactions are clinically significant. As such, more clinically relevant research in this area is necessary. This review provides information on commonly used nutrients for cardiac health and their potentials for interactions within the levels of evidence currently available.
References
- Matsudo V, Matsudo S, Andrade D, Araujo T, Andrade E, de Oliveira LC, et al. Promotion of physical activity in a developing country: The Agita Sao Paulo experience. Public Health Nutr. 2002; 5(1a): 253-261.
[Crossref] [Google Scholar] [Pubmed]
- Promotion of physical activity in a developing country: The Agita Sao Paulo experience
- Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, et al. Heart disease and stroke statistics-2016 update: A report from the American Heart Association. Circulation. 2016; 133(4): e38-e60.
- Montagnana M, Lippi G, Franchini M, Targher G, Guidi CG. Sudden cardiac death: Prevalence, pathogenesis, and prevention. Ann Med. 2008; 40(5): 360-375.
[Crossref] [Google Scholar] [Pubmed]
- Kannel WB, Thomas Jr HE. Sudden coronary death: The Framingham study. Ann N Y Acad Sci. 1982; 382(1): 3-21.
[Crossref] [Google Scholar] [Pubmed]
- Zheng ZJ, Croft JB, Giles WH, Mensah GA. Sudden cardiac death in the United States, 1989 to 1998. Circulation. 2001; 104(18): 2158-2163.
[Crossref] [Google Scholar] [Pubmed]
- Byrne R, Constant O, Smyth Y, Callagy G, Nash P, Daly K, et al. Multiple source surveillance incidence and aetiology of out-of-hospital sudden cardiac death in a rural population in the West of Ireland. Eur Heart J. 2008; 29(11): 1418-1423.
[Crossref] [Google Scholar] [Pubmed]
- McAloon CJ, Boylan LM, Hamborg T, Stallard N, Osman F, Lim PB, et al. The changing face of cardiovascular disease 2000-2012: An analysis of the world health organisation global health estimates data. Int J Cardiol. 2016; 224: 256-264.
[Crossref] [Google Scholar] [Pubmed]
- Das L, Bhaumik E, Raychaudhuri U, Chakraborty R. Role of nutraceuticals in human health. J Food Sci Technol. 2012; 49: 173-183.
[Crossref] [Google Scholar] [Pubmed]
- Muredzi ED. Food is medicine. 2013.
- Wildman RE. Handbook of nutraceuticals and functional foods. CRC press. 2016.
- Anderson KM, Castelli WP, Levy D. Cholesterol and mortality: 30 years of follow-up from the Framingham study. JAMA. 1987; 257(16): 2176-2180.
[Crossref] [Google Scholar] [Pubmed]
- Middaugh JP. Cardiovascular deaths among Alaskan Natives, 1980-86. Am J Public Health. 1990; 80(3): 282-285.
[Crossref] [Google Scholar] [Pubmed]
- Dominguez H. Functional ingredients from algae for foods and nutraceuticals. Elsevier 2013.
- Bastida S, Vaquero MP, Veldhuizen M, Sánchez‐Muniz FJ. Selected trace elements and minerals in cord blood: Association with lipids and lipoproteins at birth. Acta Paediatr. 2000; 89(10): 1201-1206.
[Crossref] [Google Scholar] [Pubmed]
- Rodenas S, Sánchez-Muniz FJ, Bastida S, Sevillano MI, Marín TL, González-Muñoz MJ. Blood pressure of omnivorous and semi-vegetarian postmenopausal women and their relationship with dietary and hair concentrations of essential and toxic metals. Nutr Hosp. 2011; 26(4): 874-883.
[Crossref] [Google Scholar] [Pubmed]
- Bibbins-Domingo K, Chertow GM, Coxson PG, Moran A, Lightwood JM, Pletcher MJ, et al. Projected effect of dietary salt reductions on future cardiovascular disease. N Engl J Med. 2010; 362(7): 590-599.
[Crossref] [Google Scholar] [Pubmed]
- He FJ, MacGregor GA. Beneficial effects of potassium. BMJ. 2001; 323(7311): 497-501.
[Crossref] [Google Scholar] [Pubmed]
- Walden R, Tomlinson B. Cardiovascular disease. 2012.
- Sesso HD, Christen WG, Bubes V, Smith JP, MacFadyen J, Schvartz M, et al. Multivitamins in the prevention of cardiovascular disease in men: The Physicians' Health Study II randomized controlled trial. JAMA. 2012; 308(17): 1751-1760.
[Crossref] [Google Scholar] [Pubmed]
- Simon JA. Vitamin C and cardiovascular disease: A review. J Am Coll Nutr. 1992;11(2):107-125.
[Crossref] [Google Scholar] [Pubmed]
- Mikirova NA, Ichim TE, Riordan NH. Anti-angiogenic effect of high doses of ascorbic acid. J Transl Med. 2008; 6(1): 50.
[Crossref] [Google Scholar] [Pubmed]
- Vinson JA, Hu SJ, Jung S, Stanski AM. A citrus extract plus ascorbic acid decreases lipids, lipid peroxides, lipoprotein oxidative susceptibility, and atherosclerosis in hypercholesterolemic hamsters. J Agric Food Chem. 1998; 46(4): 1453-1439.
- Vinson JA, Dabbagh YA, Serry MM, Jang J. Plant flavonoids, especially tea flavonols, are powerful antioxidants using an in vitro oxidation model for heart disease. J Agric Food Chem. 1995; 43(11): 2800-2802.
- Resnick LM, Müller FB, Laragh JH. Calcium-regulating hormones in essential hypertension: Relation to plasma renin activity and sodium metabolism. Ann Intern Med. 1986; 105(5): 649-654.
[Crossref] [Google Scholar] [Pubmed]
- Meagher EA, Barry OP, Lawson JA, Rokach J, FitzGerald GA. Effects of vitamin E on lipid peroxidation in healthy persons. JAMA. 2001; 285(9): 1178-1182.
[Crossref] [Google Scholar] [Pubmed]
- Weickert MO, Pfeiffer AF. Metabolic effects of dietary fiber consumption and prevention of diabetes. J Nutr. 2008; 138(3): 439-442.
[Crossref] [Google Scholar] [Pubmed]
- Ullah MF, Khan MW. Food as medicine: Potential therapeutic tendencies of plant derived polyphenolic compounds. Asian Pac J Cancer Prev. 2008; 9(2): 187-196.
[Google Scholar] [Pubmed]
- Maxwell S, Cruickshank A, Thorpe G. Red wine and antioxidant activity in serum. Lancet. 1994; 344(8916): 193-194.
[Crossref] [Google Scholar] [Pubmed]
- Gresele P, Pignatelli P, Guglielmini G, Carnevale R, Mezzasoma AM, Ghiselli A, et al. Resveratrol, at concentrations attainable with moderate wine consumption, stimulates human platelet nitric oxide production. J Nutr. 2008; 138(9): 1602-1608.
[Crossref] [Google Scholar] [Pubmed]
- Das DK, Mukherjee S, Ray D. Erratum to: Resveratrol and red wine, healthy heart and longevity. Heart Fail Rev. 2011; 16: 425-435.
[Crossref] [Google Scholar] [Pubmed]
- Buijsse B, Weikert C, Drogan D, Bergmann M, Boeing H. Chocolate consumption in relation to blood pressure and risk of cardiovascular disease in German adults. Eur Heart J. 2010; 31(13): 1616-1623.
[Crossref] [Google Scholar] [Pubmed]
- Erkkilä AT, Booth SL. Vitamin K intake and atherosclerosis. Curr Opin Lipidol. 2008; 19(1): 39-42.
[Crossref] [Google Scholar] [Pubmed]
- Houston MC, Harper KJ. Potassium, magnesium, and calcium: Their role in both the cause and treatment of hypertension. J Clin Hypertens. 2008; 10(7): 3-11.
[Crossref] [Google Scholar] [Pubmed]
- Shechter M, Merz CN, Paul-Labrador M, Meisel SR, Rude RK, Molloy MD, et al. Oral magnesium supplementation inhibits platelet-dependent thrombosis in patients with coronary artery disease. Am J Cardiol. 1999; 84(2): 152-156.
[Crossref] [Google Scholar] [Pubmed]
- Anker SD, Comin Colet J, Filippatos G, Willenheimer R, Dickstein K, Drexler H, et al. Ferric carboxymaltose in patients with heart failure and iron deficiency. N Engl J Med. 2009; 361(25): 2436-2348.
[Crossref] [Google Scholar] [Pubmed]
- Hummel M, Standl E, Schnell O. Chromium in metabolic and cardiovascular disease. Horm Metab Res. 2007; 39(10): 743-751.
[Crossref] [Google Scholar] [Pubmed]
- Schroeder HA. Cadmium, chromium, and cardiovascular disease. Circulation. 1967; 35(3): 570-582.
[Crossref] [Google Scholar] [Pubmed]
- Balk EM, Tatsioni A, Lichtenstein AH, Lau J, Pittas AG. Effect of chromium supplementation on glucose metabolism and lipids: A systematic review of randomized controlled trials. Diabetes Care. 2007; 30(8): 2154-2163.
[Crossref] [Google Scholar] [Pubmed]
- Hercberg S, Galan P, Preziosi P, Bertrais S, Mennen L, Malvy D, et al. The SU VI. MAX Study: A randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch Intern Med. 2004; 164(21): 2335-2342.
[Crossref] [Google Scholar] [Pubmed]
- Breslow JL. n-3 Fatty acids and cardiovascular disease. Am J Clin Nutr. 2006; 83(6): 1477S-1482S.
[Crossref] [Google Scholar] [Pubmed]
- León H, Shibata MC, Sivakumaran S, Dorgan M, Chatterley T, Tsuyuki RT. Effect of fish oil on arrhythmias and mortality: Systematic review. BMJ. 2008; 337.
[Crossref] [Google Scholar] [Pubmed]
- Greenberg ER, Baron JA, Karagas MR, Stukel TA, Nierenberg DW, Stevens MM, et al. Mortality associated with low plasma concentration of beta carotene and the effect of oral supplementation. JAMA. 1996; 275(9): 699-703.
[Crossref] [Google Scholar] [Pubmed]
- Werman MJ, Sukenik A, Mokady S. Effects of the marine unicellular alga Nannochloropsis sp. to reduce the plasma and liver cholesterol levels in male rats fed on diets with cholesterol. Biosci Biotechnol Biochem. 2003; 67(10): 2266-2268.
[Crossref] [Google Scholar] [Pubmed]
- Athukorala Y, Ahn GN, Jee YH, Kim GY, Kim SH, Ha JH, et al. Antiproliferative activity of sulfated polysaccharide isolated from an enzymatic digest of Ecklonia cava on the U-937 cell line. J Appl Phycol. 2009; 21: 307-314.
- Nishino T, Nishioka C, Ura H, Nagumo T. Isolation and partial characterization of a noval amino sugar-containing fucan sulfate from commercial Fucus vesiculosus fucoidan. Carbohydr Res. 1994; 255: 213-224.
[Crossref] [Google Scholar] [Pubmed]
- Chevolot L, Foucault A, Chaubet F, Kervarec N, Sinquin C, Fisher AM, et al. Further data on the structure of brown seaweed fucans: Relationships with anticoagulant activity. Carbohydr Res. 1999; 319(1-4): 154-165.
[Crossref] [Google Scholar] [Pubmed]
- Murata M, Nakazoe JI. Production and use of marine aIgae in Japan. Japan Agric Res Q. 2001; 35(4): 281-290.
- Shanmugam M, Mody KH. Heparinoid-active sulphated polysaccharides from marine algae as potential blood anticoagulant agents. Curr Sci. 2000: 1672-1683.
- Noda H. Health benefits and nutritional properties of nori. J Appl Phycol. 1993; 5: 255-258.
- Sato M, Hosokawa T, Yamaguchi T, Nakano T, Muramoto K, Kahara T, et al. Angiotensin I-converting enzyme inhibitory peptides derived from wakame (Undaria pinnatifida) and their antihypertensive effect in spontaneously hypertensive rats. J Agric Food Chem. 2002; 50(21): 6245-6252.
[Crossref] [Google Scholar] [Pubmed]
- Mendis E, Kim SK. Present and future prospects of seaweeds in developing functional foods. Adv Food Nutr Res. 2011; 64: 1-5.
[Crossref] [Google Scholar] [Pubmed]
- Tufail T, Saeed F, Abbas M, Arshad MU, Nadeem MT, Ain HB, et al. Marine bioactives: Potentials to reduce the incidence of cardiovascular disorders. Curr Top Nutraceutical Res. 2018; 16(1).
- Mukhtar H, Ahmad N. Tea polyphenols: Prevention of cancer and optimizing health. Am J Clin Nutr. 2000; 71(6): 1698S-1702S.
[Crossref] [Google Scholar] [Pubmed]
- Dewell A, Hollenbeck PL, Hollenbeck CB. A critical evaluation of the role of soy protein and isoflavone supplementation in the control of plasma cholesterol concentrations. J Clin Endocrinol Metab. 2006; 91(3): 772-780.
[Crossref] [Google Scholar] [Pubmed]
- Rowland I, Faughnan M, Hoey L, Wähälä K, Williamson G, Cassidy A. Bioavailability of phyto-oestrogens. Br J Nutr. 2003; 89(S1): S45-S58.
[Crossref] [Google Scholar] [Pubmed]
- Weber C, Bysted A, Hølmer G. Coenzyme Q10 in the diet-daily intake and relative bioavailability. Mol Aspects Med. 1997; 18: 251-254.
[Crossref] [Google Scholar] [Pubmed]
- Persky AM, Brazeau GA. Clinical pharmacology of the dietary supplement creatine monohydrate. Pharmacol Rev. 2001; 53(2): 161-176.
[Google Scholar] [Pubmed]
- Kletzmayr J, Mayer G, Legenstein E, Heinz-Peer G, Leitha T, Hörl WH, et al. Anemia and carnitine supplementation in hemodialyzed patients. Kidney Int Suppl. 1999; 55: S93-S106.
[Crossref] [Google Scholar] [Pubmed]
- Diwadkar-Navsariwala V, Novotny JA, Gustin DM, Sosman JA, Rodvold KA, Crowell JA, et al. A physiological pharmacokinetic model describing the disposition of lycopene in healthy men. J Lipid Res. 2003; 44(10): 1927-1939.
[Crossref] [Google Scholar] [Pubmed]
- Alves-Rodrigues A, Shao A. The science behind lutein. Toxicol Lett. 2004; 150(1): 57-83.
[Crossref] [Google Scholar] [Pubmed]
- Pepping J. DHEA: Dehydroepiandrosterone. Am J Health Syst Pharm. 2000; 57(22): 2048-2056.
[Crossref] [Google Scholar] [Pubmed]
- Lin Y, Rudrum M, van der Wielen RP, Trautwein EA, McNeill G, Sierksma A, et al. Wheat germ policosanol failed to lower plasma cholesterol in subjects with normal to mildly elevated cholesterol concentrations. Metabolism. 2004; 53(10): 1309-1314.
[Crossref] [Google Scholar] [Pubmed]
- Düweler KG, Rohdewald P. Urinary metabolites of French maritime pine bark extract in humans. Pharmazie. 2000; 55(5): 364-368.
[Google Scholar] [Pubmed]
- Wolter F, Stein J. Biological activities of resveratrol and its analogs. Drugs Future. 2002; 27(10): 949-959.
- Ward NC, Croft KD, Puddey IB, Hodgson JM. Supplementation with grape seed polyphenols results in increased urinary excretion of 3-hydroxyphenylpropionic acid, an important metabolite of proanthocyanidins in humans. J Agric Food Chem. 2004; 52(17): 5545-5549.
[Crossref] [Google Scholar] [Pubmed]
- Donovan JL, Chavin KD, Devane CL, Taylor RM, Wang JS, Ruan Y, et al. Green tea (Camellia sinensis) extract does not alter cytochrome p450 3A4 or 2D6 activity in healthy volunteers. Drug Metab Dispos. 2004; 32(9): 906-908.
[Crossref] [Google Scholar] [Pubmed]
- Hodgson JM, Puddey IB, Burke V, Beilin LJ, Mori TA, Chan SY. Acute effects of ingestion of black tea on postprandial platelet aggregation in human subjects. Br J Nutr. 2002; 87(2): 141-145.
[Crossref] [Google Scholar] [Pubmed]
- Chow HS, Hakim IA, Vining DR, Crowell JA, Cordova CA, Chew WM, et al. Effects of repeated green tea catechin administration on human cytochrome P450 activity. Cancer Epidemiol Biomarkers Prev. 2006; 15(12): 2473-2476.
[Crossref] [Google Scholar] [Pubmed]
- Zhong Z, Connor HD, Li X, Mason RP, Forman DT, Lemasters JJ, Thurman RG. Reduction of ciclosporin and tacrolimus nephrotoxicity by plant polyphenols. J Pharm Pharmacol. 2006; 58(11): 1533-1543.
[Crossref] [Google Scholar] [Pubmed]
- Greenblatt DJ, von Moltke LL, Perloff ES, Luo Y, Harmatz JS, Zinny MA. Interaction of flurbiprofen with cranberry juice, grape juice, tea, and fluconazole: In vitro and clinical studies. Clin Pharmacol Ther. 2006; 79(1): 125-133.
[Crossref] [Google Scholar] [Pubmed]
- Dai YH, Wei JR, Chen XQ. Interactions between tea polyphenols and nutrients in food. Compr Rev Food Sci Food Saf. 2023.
[Crossref] [Google Scholar] [Pubmed]
- Lambert JD, Hong J, Kim DH, Mishin VM, Yang CS. Piperine enhances the bioavailability of the tea polyphenol (−)-epigallocatechin-3-gallate in mice. J Nutr. 2004; 134(8): 1948-1952.
[Crossref] [Google Scholar] [Pubmed]
- Mirkov S, Komoroski BJ, Ramírez J, Graber AY, Ratain MJ, Strom SC, et al. Effects of green tea compounds on irinotecan metabolism. Drug Metab Dispos. 2007; 35(2): 228-233.
[Crossref] [Google Scholar] [Pubmed]
- Zijp IM, Korver O, Tijburg LB. Effect of tea and other dietary factors on iron absorption. Crit Rev Food Sci Nutr. 2000; 40(5): 371-398.
[Crossref] [Google Scholar] [Pubmed]
- Parker DL, Hoffmann TK, Tucker MA, Meier DJ. Interaction between warfarin and black tea. Ann Pharmacother. 2009; 43(1): 150-151.
[Crossref] [Google Scholar] [Pubmed]
- Shaikh AS, Thomas AB, Chitlange SS. Herb-drug interaction studies of herbs used in treatment of cardiovascular disorders-A narrative review of preclinical and clinical studies. Phytother Res. 2020; 34(5): 1008-1026.
[Crossref] [Google Scholar] [Pubmed]
- Peng SX, Ritchie DM, Cousineau M, Danser E, deWire R, Floden J. Altered oral bioavailability and pharmacokinetics of P-glycoprotein substrates by coadministration of biochanin A. J Pharm Sci. 2006; 95(9): 1984-1993.
[Crossref] [Google Scholar] [Pubmed]
- Nakajima M, Itoh M, Yamanaka H, Fukami T, Tokudome S, Yamamoto Y, et al. Isoflavones inhibit nicotine C‐oxidation catalyzed by human CYP2A6. The J Clin Pharmacol. 2006; 46(3): 337-344.
[Crossref] [Google Scholar] [Pubmed]
- Li X, Choi JS. Effect of genistein on the pharmacokinetics of paclitaxel administered orally or intravenously in rats. Int J Pharm. 2007; 337(1-2): 188-193.
[Crossref] [Google Scholar] [Pubmed]
- Messina MJ, Loprinzi CL. Soy for breast cancer survivors: A critical review of the literature. J Nutr. 2001; 131(11): 3095S-3108S.
[Crossref] [Google Scholar] [Pubmed]
- Monostory K, Vereczkey L. The effect of ipriflavone and its main metabolites on theophylline biotransformation. Eur J Drug Metab Pharmacokinet. 1996; 21: 61-66.
[Crossref] [Google Scholar] [Pubmed]
- Prasad K. Flaxseed and cardiovascular health. J Cardiovasc Pharmacol. 2009; 54(5): 369-377.
[Crossref] [Google Scholar] [Pubmed]
- Lemay A, Dodin S, Kadri N, Jacques H, Forest JC. Flaxseed dietary supplement versus hormone replacement therapy in hypercholesterolemic menopausal women. Obstet Gynecol. 2002; 100(3): 495-504.
[Crossref] [Google Scholar] [Pubmed]
- Itagaki S, Kurokawa T, Nakata C, Saito Y, Oikawa S, Kobayashi M, et al. In vitro and in vivo antioxidant properties of ferulic acid: A comparative study with other natural oxidation inhibitors. Food Chem. 2009; 114(2): 466-471.
- Badmaev V, Majeed M, Prakash L. Piperine derived from black pepper increases the plasma levels of coenzyme Q10 following oral supplementation. J Nutr Biochem. 2000; 11(2): 109-113.
[Crossref] [Google Scholar] [Pubmed]
- Landbo C, Almdal TP. Interaction between warfarin and coenzyme Q10. Ugeskr Laeger. 1998; 160(22): 3226-3227.
[Google Scholar] [Pubmed]
- Igarashi T, Kobayashi M, Sugiyama K, Sagami F, Ohtake S. Effect of coenzyme Q10 on electrolyte metabolism and the interaction with aldosterone in rats and dogs. Proc West Pharmacol Soc. 1975; 18: 399-402.
[Google Scholar] [Pubmed]
- Zhou Q, Chowbay B. Effect of coenzyme Q 10 on the disposition of doxorubicin in rats. Eur J Drug Metab Pharmacokinet. 2002; 27(3): 185-192.
[Crossref] [Google Scholar] [Pubmed]
- Vandenberghe K, Gillis N, van Leemputte M, van Hecke P, Vanstapel F, Hespel P. Caffeine counteracts the ergogenic action of muscle creatine loading. J Appl Physiol. 1996; 80(2): 452-457.
[Crossref] [Google Scholar] [Pubmed]
- Steenge GR, Simpson EJ, Greenhaff PL. Protein-and carbohydrate-induced augmentation of whole body creatine retention in humans. J Appl Physiol. 2000; 89(3): 1165-1171.
[Crossref] [Google Scholar] [Pubmed]
- Ahuja KD, Ashton EL, Ball MJ. Effects of a high monounsaturated fat, tomato-rich diet on serum levels of lycopene. Eur J Clin Nutr. 2003; 57(7): 832-841.
[Crossref] [Google Scholar] [Pubmed]
- Johnson EJ, Qin J, Krinsky NI, Russell RM. Ingestion by men of a combined dose of β-carotene and lycopene does not affect the absorption of β-carotene but improves that of lycopene. J Nutr. 1997; 127(9): 1833-1837.
[Crossref] [Google Scholar] [Pubmed]
- Elinder LS, Hådell K, Johansson J, Mølgaard J, Holme I, Olsson AG, et al. Probucol treatment decreases serum concentrations of diet-derived antioxidants. Arterioscler Thromb Vasc Biol. 1995; 15(8): 1057-1063.
[Crossref] [Google Scholar] [Pubmed]
- Koonsvitsky BP, Berry DA, Jones MB, Lin PY, Cooper DA, Jones DY, et al. Olestra affects serum concentrations of α-tocopherol and carotenoids but not vitamin D or vitamin K status in free-living subjects. J Nutr. 1997; 127(8): 1636S-1645S.
[Crossref] [Google Scholar] [Pubmed]
- Carbajal D, Arruzazabala ML, Valdés S, Más D. Interaction policosanol-warfarin on bleeding time and thrombosis in rats. Pharmacol Res. 1998; 38(2): 89-91.
[Crossref] [Google Scholar] [Pubmed]
- Castano G, Mas R, Gamez R, Fernandez J, Illnait J, Fernandez L, et al. Concomitant use of policosanol and beta-blockers in older patients. Int J Clin Pharmacol Res. 2004; 24(2-3): 65-77.
[Google Scholar] [Pubmed]
- Pérez-Souto N, Acosta PC, Mederos CM, Reyes JL, Martínez O. Efecto Del Ateromixol (PPG) Sobre La Farmacocinetica De La Anti Pi Rina. Revista CENIC Ciencias Biológicas. 1991; 22(1-2): 077-078.
- Arruzazabala ML, Carbajal D, Más R, Valdés S, Molina V. Pharmacological interaction between policosanol and nitroprusside in rats. J Med Food. 2001; 4(2): 67-70.
[Crossref] [Google Scholar] [Pubmed]
- Otmani S, Demazieres A, Staner C, Jacob N, Nir T, Zisapel N, et al. Effects of prolonged‐release melatonin, zolpidem, and their combination on psychomotor functions, memory recall, and driving skills in healthy middle aged and elderly volunteers. Hum Psychopharmacol. 2008; 23(8): 693-705.
[Crossref] [Google Scholar] [Pubmed]
- Golombek DA, Escolar E, Burin LJ, de Brito Sánchez MG, Duque DF, Cardinali DP. Chronopharmacology of melatonin: Inhibition by benzodiazepine antagonism. Chronobiol Int. 1992; 9(2): 124-131.
[Crossref] [Google Scholar] [Pubmed]
- Obochi GO, Amali OO, Ochalefu DO. Effect of melatonin and caffeine interaction on caffeine induced oxidative stress and sleep disorders. Niger J Physiol Sci. 2010; 25(1): 17-24.
[Google Scholar] [Pubmed]
- Gupta M, Gupta YK, Agarwal S, Aneja S, Kalaivani M, Kohli K. Effects of add‐on melatonin administration on antioxidant enzymes in children with epilepsy taking carbamazepine monotherapy: A randomized, double‐blind, placebo‐controlled trial. Epilepsia. 2004; 45(12): 1636-1639.
[Crossref] [Google Scholar] [Pubmed]
- Lusardi P, Piazza E, Fogari R. Cardiovascular effects of melatonin in hypertensive patients well controlled by nifedipine: A 24‐hour study. Br J Clin Pharmacol. 2000; 49(5): 423-427.
[Crossref] [Google Scholar] [Pubmed]
- Hilli J, Korhonen T, Turpeinen M, Hokkanen J, Mattila S, Laine K. The effect of oral contraceptives on the pharmacokinetics of melatonin in healthy subjects with CYP1A2 g.‐163C>A polymorphism. J Clin Pharmacol. 2008; 48(8): 986-994.
[Crossref] [Google Scholar] [Pubmed]
- Turkistani A, Abdullah KM, Al-Shaer AA, Mazen KF, Alkatheri K. Melatonin premedication and the induction dose of propofol. Eur J Anaesthesiol. 2007; 24(5): 399-402.
[Crossref] [Google Scholar] [Pubmed]
- Souêtre E, Salvati E, Belugou JL, Pringuey D, Candito M, Krebs B, et al. Circadian rhythms in depression and recovery: Evidence for blunted amplitude as the main chronobiological abnormality. Psychiatry Res. 1989; 28(3): 263-278.
[Crossref] [Google Scholar] [Pubmed]
- Foster BC, Cvijovic K, Boon HS, Tam TW, Liu R, Murty M, et al. Melatonin interaction resulting in severe sedation. J Pharm Pharm Sci. 2015; 18(2): 124-131.
[Crossref] [Google Scholar] [Pubmed]
- Ursing C, Bahr CV, Brismar K, Röjdmark S. Influence of cigarette smoking on melatonin levels in man. Eur J Clin Pharmacol. 2005; 61: 197-201.
[Crossref] [Google Scholar] [Pubmed]
- Herxheimer A, Petrie KJ, Cochrane Common Mental Disorders Group. Melatonin for the prevention and treatment of jet lag. Cochrane Database Syst Rev. 1996; 2010(1).
[Crossref] [Google Scholar] [Pubmed]
- Stef G, Csiszar A, Lerea K, Ungvari Z, Veress G. Resveratrol inhibits aggregation of platelets from high-risk cardiac patients with aspirin resistance. J Cardiovasc Pharmacol. 2006; 48(2): 1-5.
[Crossref] [Google Scholar] [Pubmed]
- Yu C, Shin YG, Kosmeder JW, Pezzuto JM, van Breemen RB. Liquid chromatography/tandem mass spectrometric determination of inhibition of human cytochrome P450 isozymes by resveratrol and resveratrol‐3‐sulfate. Rapid Commun Mass Spectrom. 2003; 17(4): 307-313.
[Crossref] [Google Scholar] [Pubmed]
- Václavíková R, Horský S, Šimek P, Gut I. Paclitaxel metabolism in rat and human liver microsomes is inhibited by phenolic antioxidants. Naunyn Schmiedebergs Arch Pharmacol. 2003; 368: 200-209.
[Crossref] [Google Scholar] [Pubmed]
- Ward NC, Hodgson JM, Croft KD, Burke V, Beilin LJ, Puddey IB. The combination of vitamin C and grape-seed polyphenols increases blood pressure: A randomized, double-blind, placebo-controlled trial. J Hypertens. 2005; 23(2): 427-434.
[Crossref] [Google Scholar] [Pubmed]
- Nishikawa M, Ariyoshi N, Kotani A, Ishii I, Nakamura H, Nakasa H, et al. Effects of continuous ingestion of green tea or grape seed extracts on the pharmacokinetics of midazolam. Drug Metab Pharmacokinet. 2004; 19(4): 280-289.
[Crossref] [Google Scholar] [Pubmed]
- Bailey DG, Dresser GK, Leake BF, Kim RB. Naringin is a major and selective clinical inhibitor of organic anion‐transporting polypeptide 1A2 (OATP1A2) in grapefruit juice. Clin Pharmacol Ther. 2007; 81(4): 495-502.
[Crossref] [Google Scholar] [Pubmed]
- Sica DA. Interaction of grapefruit juice and calcium channel blockers. Am J Hypertens. 2006; 19(7): 768-773.
[Crossref] [Google Scholar] [Pubmed]
- Peynaud D, Charpiat B, Vial T, Gallavardin M, Ducerf C. Tacrolimus severe overdosage after intake of masked grapefruit in orange marmalade. Eur J Clin Pharmacol. 2007; 63(7): 721-722.
[Crossref] [Google Scholar] [Pubmed]
- Brandin H, Myrberg O, Rundlöf T, Arvidsson AK, Brenning G. Adverse effects by artificial grapefruit seed extract products in patients on warfarin therapy. Eur J Clin Pharmacol. 2007; 63: 565-570.
[Crossref] [Google Scholar] [Pubmed]
Author Info
Jitendra Singh Yadav1, Sonia Pandey2 and Arti Gupta1*2Department of Pharmaceutical Sciences, Rama University, Uttar Pradesh, India
Citation: Yadav JS: Role of Essential Nutrients for Cardiovascular Health: Risk and Management of Drug Interaction
Received: 12-May-2023 Accepted: 26-May-2023 Published: 02-Jun-2023, DOI: 10.31858/0975-8453.14.6.418-430
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
ARTICLE TOOLS
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Psychometric properties of the World Health Organization Quality of life instrument, short form: Validity in the Vietnamese healthcare context Trung Quang Vo*, Bao Tran Thuy Tran, Ngan Thuy Nguyen, Tram ThiHuyen Nguyen, Thuy Phan Chung Tran SRP. 2020; 11(1): 14-22 » doi: 10.5530/srp.2019.1.3
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- Deuterium Depleted Water as an Adjuvant in Treatment of Cancer Anton Syroeshkin, Olga Levitskaya, Elena Uspenskaya, Tatiana Pleteneva, Daria Romaykina, Daria Ermakova SRP. 2019; 10(1): 112-117 » doi: 10.5530/srp.2019.1.19
- Dental Development between Assisted Reproductive Therapy (Art) and Natural Conceived Children: A Comparative Pilot Study Norzaiti Mohd Kenali, Naimah Hasanah Mohd Fathil, Norbasyirah Bohari, Ahmad Faisal Ismail, Roszaman Ramli SRP. 2020; 11(1): 01-06 » doi: 10.5530/srp.2020.1.01
- Manilkara zapota (L.) Royen Fruit Peel: A Phytochemical and Pharmacological Review Karle Pravin P, Dhawale Shashikant C SRP. 2019; 10(1): 11-14 » doi: 0.5530/srp.2019.1.2
- Pharmacognostic and Phytopharmacological Overview on Bombax ceiba Pankaj Haribhau Chaudhary, Mukund Ganeshrao Tawar SRP. 2019; 10(1): 20-25 » doi: 10.5530/srp.2019.1.4
- A Review of Pharmacoeconomics: the key to “Healthcare for All” Hasamnis AA, Patil SS, Shaik Imam, Narendiran K SRP. 2019; 10(1): s40-s42 » doi: 10.5530/srp.2019.1s.21
- A Prospective Review on Phyto-Pharmacological Aspects of Andrographis paniculata Govindraj Akilandeswari, Arumugam Vijaya Anand, Palanisamy Sampathkumar, Puthamohan Vinayaga Moorthi, Basavaraju Preethi SRP. 2019; 10(1): 15-19 » doi: 10.5530/srp.2019.1.3






